NMPA gives emergency approvals to COVID-19 test kits
The National Medical Products Administration (NMPA) recently approved a rapid nucleic acid reagent test kit for COVID-19 to further meet the needs of epidemic prevention and control.
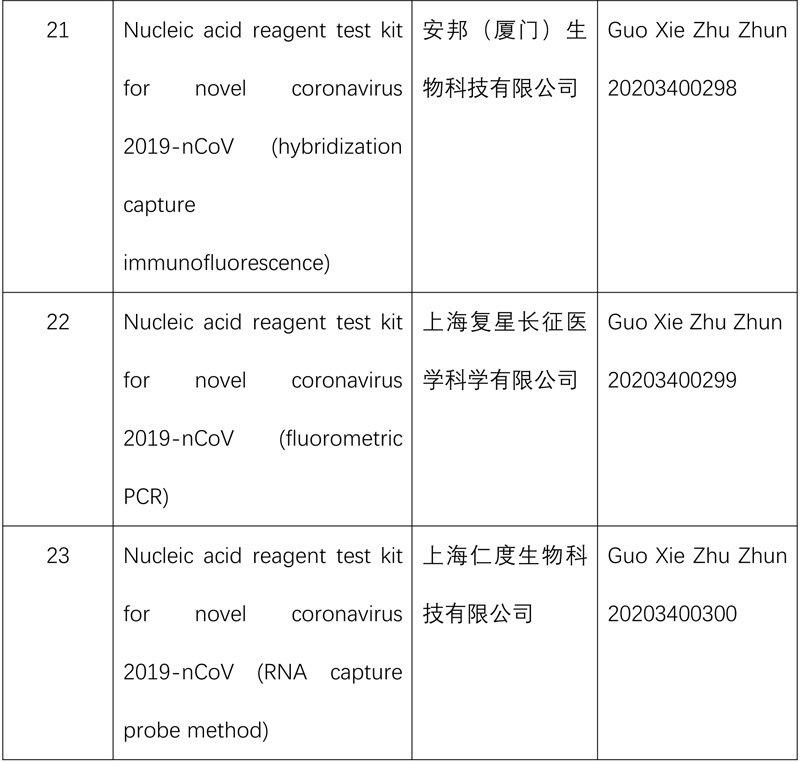
The product adopts RNA specific target capture and real-time transcription-mediated constant temperature amplification detection technology, which enables nucleic acid extraction and amplification to be completed automatically in a reaction tube.
The results can be obtained in 90 minutes, and continuous parallel testing can be realized to improve its efficiency. Relevant data show that the sensitivity and specificity of the results are equivalent to the performance of the traditional polymerase chain reaction (PCR) method.
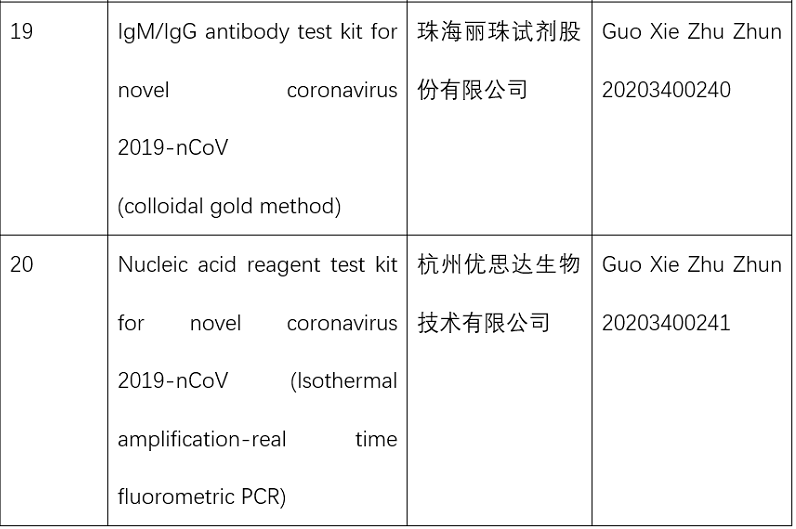
Before this emergency approval, the NMPA had approved three rapid nucleic acid test kits, one with isothermal amplification-real time fluorometric PCR and two using the hybridization capture immunofluorescence method.
These products shortened detection time, improved efficiency, and served the need of epidemic prevention and control.
The NMPA also issued emergency approval for a nucleic acid reagent test kit (fluorescence PCR) developed by a Shanghai enterprise to further expand the supply of detection reagents for epidemic prevention and control.
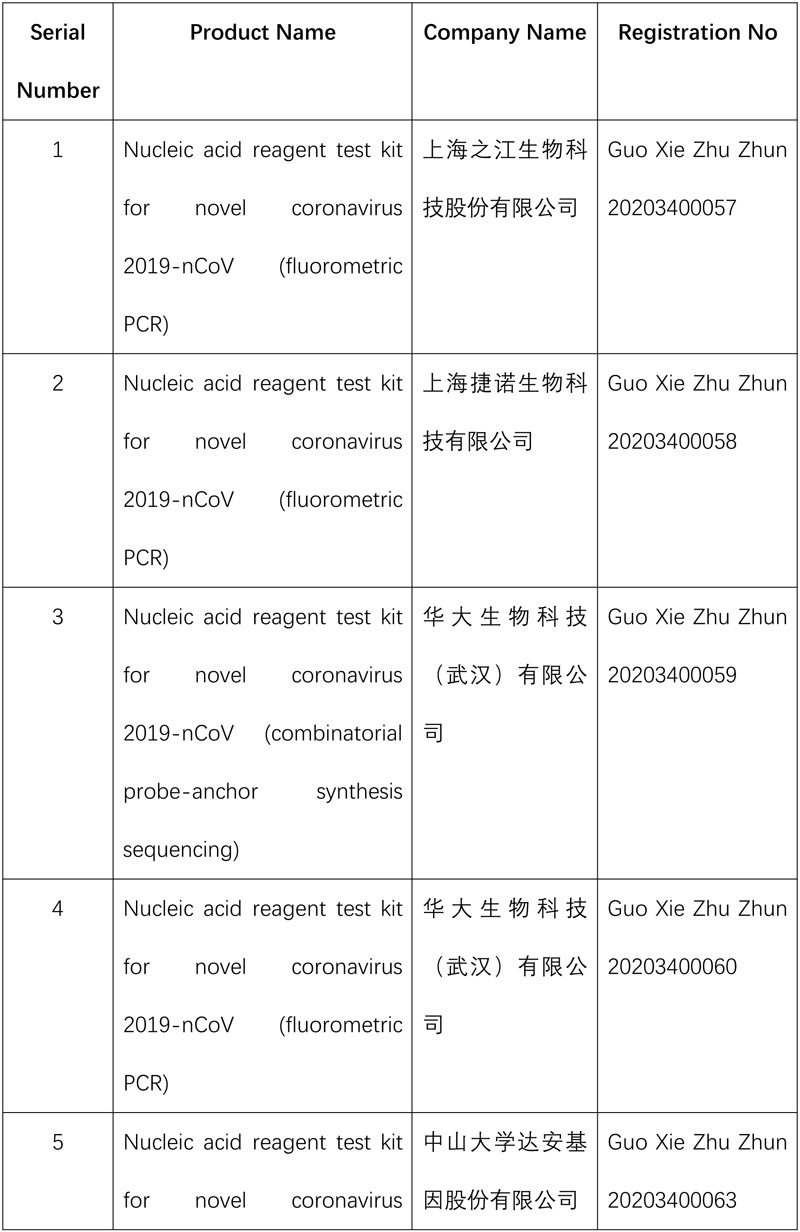
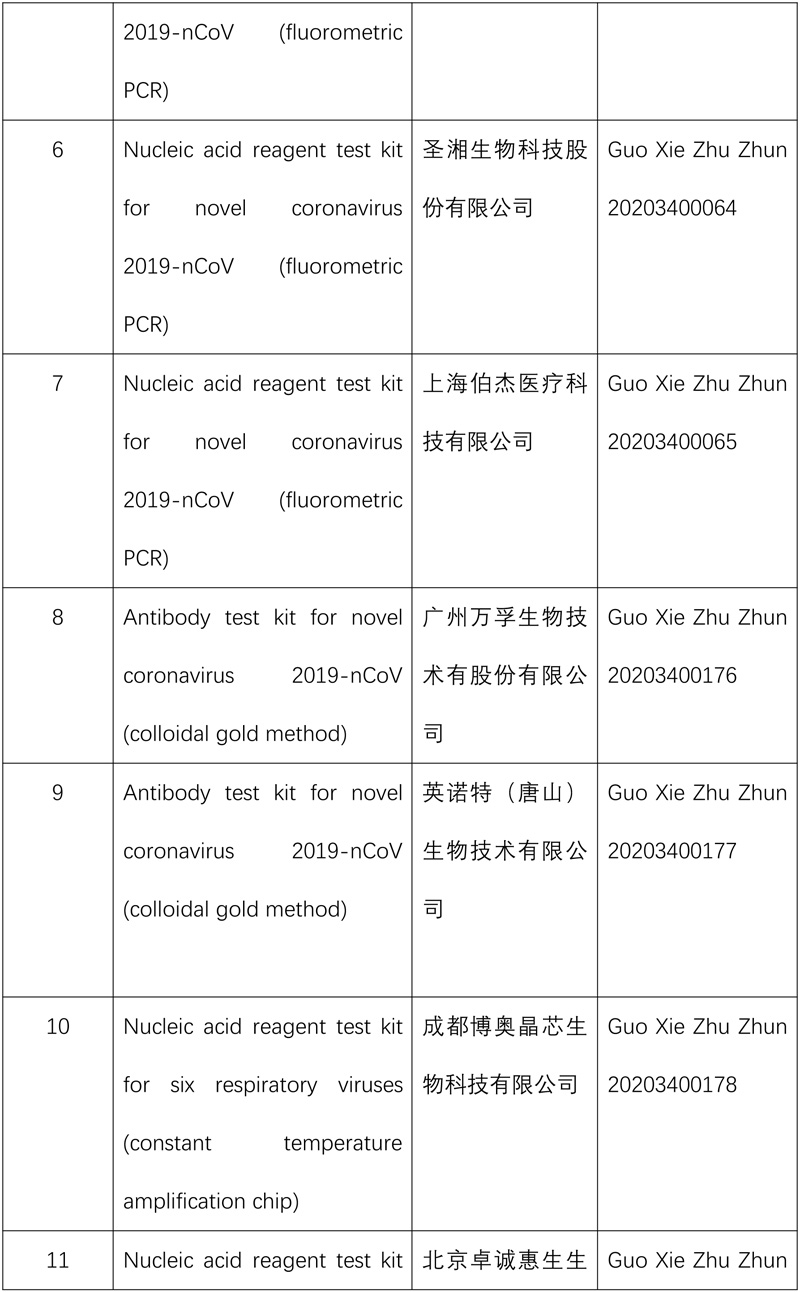
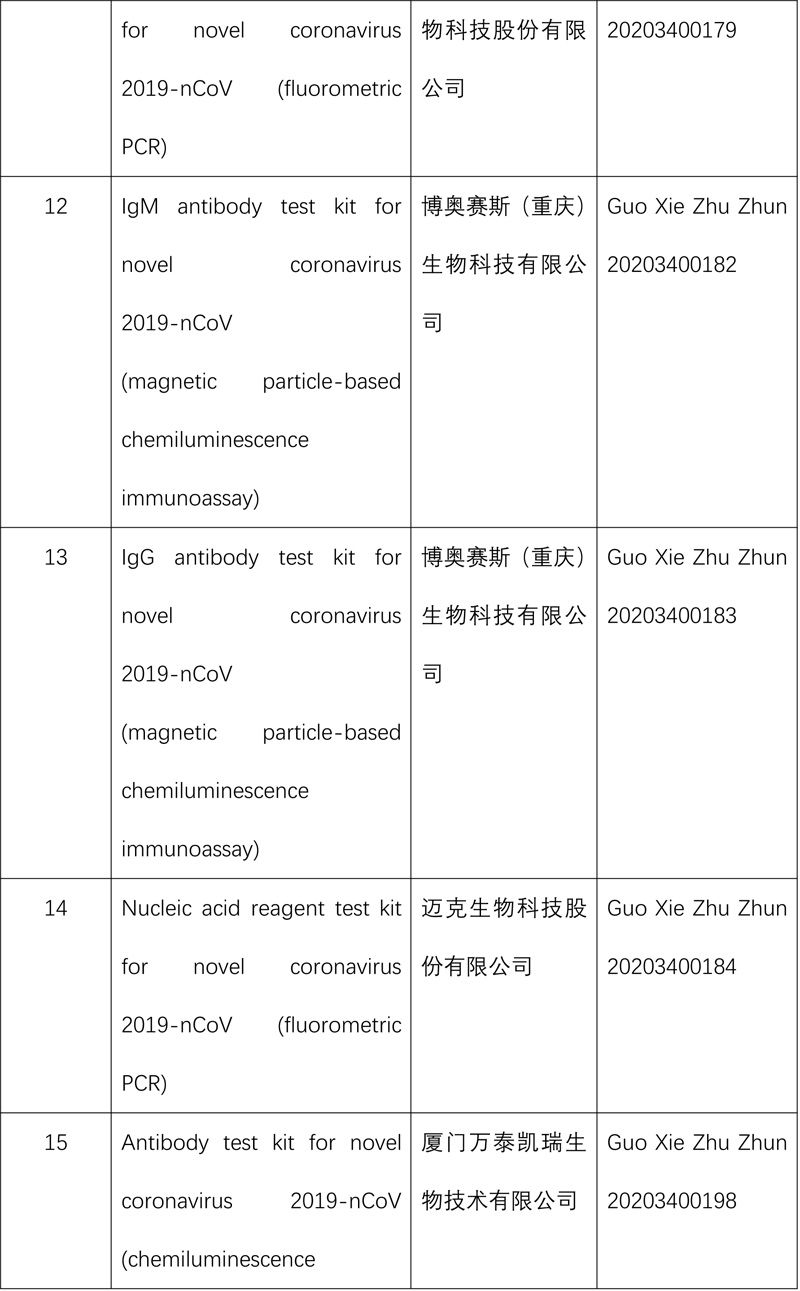
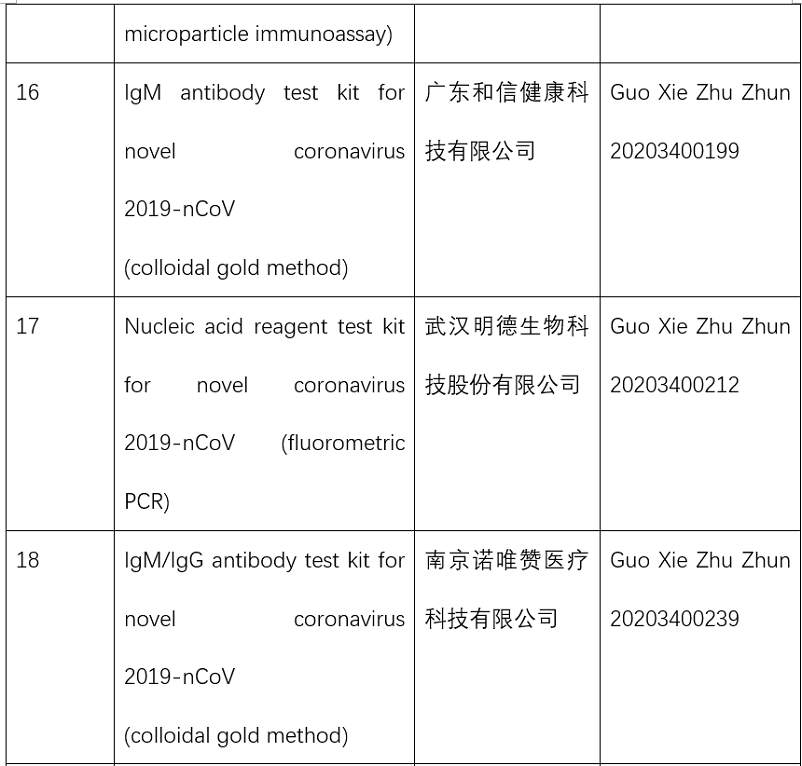
So far, the NMPA has approved 15 nucleic acid reagent test kits and eight antibody reagent test kits for COVID-19.